Researchers at MRC Harwell, alongside their colleagues at Université de Strasbourg and Great Ormond Street Institute of Child Health, have published a review in Molecular Therapy. The review explores the reliability and challenges of genome editing application in both research and in clinic.
Genome editing (GE) has revolutionised biomedical research. GE tools induce a break in the double-stranded DNA which gives researchers the ability to add, remove or alter the genetic material of an organism. These tools are promising as they are currently being developed for potential therapies for a number of diseases. However, the precision of these tools remains uncertain and there are a range of challenges which need to be considered before they are widely used in clinic.
This review paper identified the various challenges and risks associated with GE and discussed their implications and potential solutions.
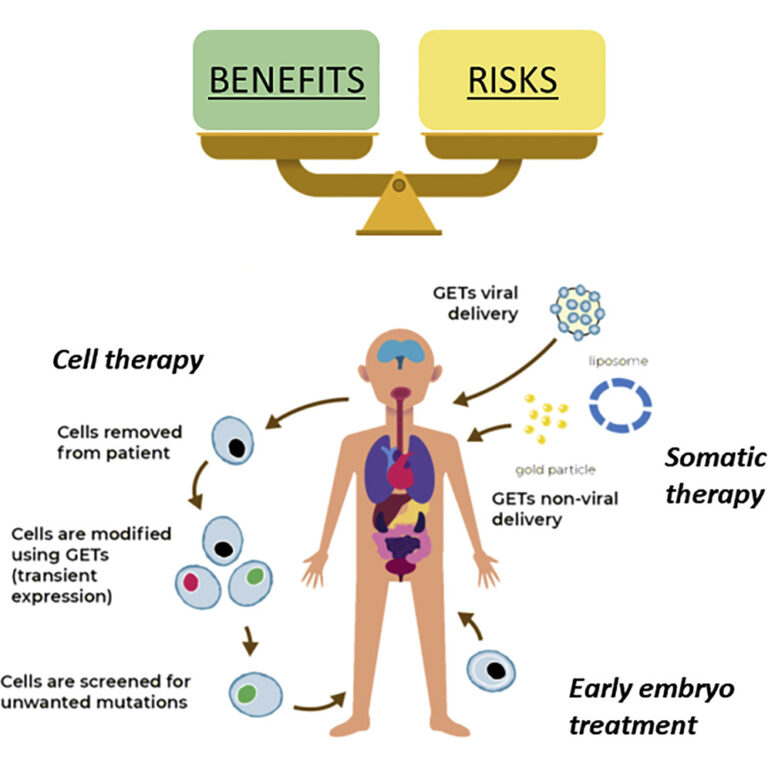
The paper discussed a benefit:risk threshold; identifying that the benefits of a cure or treatment need to be considered alongside a risk analysis before clinical practices can be safe. The risks associated were identified as the potential for on and off target genetic alterations, the risks associated with various delivery methods and the difference in outcomes of GE across various contexts, for example, different cell types.
The risks associated with this mechanism include off-target effects as well as, and more commonly, local genetic rearrangement at on-target sites, which could result in various new mutations. The unwanted consequences of using these tools need to be assessed in research across various contexts in order to ensure safety and guide their use in clinic.
In addition, the review also discussed the impact of the context of application and delivery mechanism as an important factor to consider. Each combination of context and delivery mechanism pose their own challenges but sometimes also offer solutions, and the safety of each must be validated before use in a clinical setting. The variability of GE across cell and organ types can be assessed within research to inform their use in clinics, however, clinical trials are also required for full assessment of the reliability of GE therapies.
Lydia Teboul, an author on the paper, comments:
“The initial expectation for Genome Editing as a perfectly precise technology had placed the bar at an impossibly high level. We cannot decide the risk attached to any category of genome editing tools as a whole. Each design needs to be evaluated for each intervention, whether it is as a research model or in clinic.”