Mammalian Genetics Unit PhD student Paige Street is commended for their article, Schizophrenia – the gene ‘keeping it in the family’
Paige Street, a PhD student in the Neurobehavioural Genetics group at MRC Harwell, has received a commendation for their article submitted to the MRC’s 2021 Max Perutz Science Writing Award. The award, named after Nobel Laureate Max Perutz, aims to encourage and recognise outstanding articles by MRC PhD students that engage a general audience and entries are judged on creativity, content, structure, and timeliness.
About receiving this recognition, Paige commented, “I’m completely chuffed to win the commendation prize; it’s great to have your work recognised. I definitely feel encouraged to engage in science communication work in the future!”
Here is Paige’s article:
Schizophrenia – the gene ‘keeping it in the family’
At the end of the day, you retreat into the safe sanctuary of home. Suddenly, you hear knocking at the door. You answer, but you can’t see anyone outside. You grumble about neighbourhood youths, and go back indoors. Soon you hear it again – louder than before. The doorway is still empty when you yank open the door, the pranksters nowhere to be found.
You ignore the further knocking – but then you start to feel their eyes watching you through the curtains, and see threatening shadows moving outside. You’ve been seeing and hearing these tormenters for years, following you wherever you go. Your mum sees these monsters, and so did her dad. There is a chance your children will be haunted, too.
Schizophrenia is a psychiatric illness affecting 1 in 100 people in the UK. A schizophrenic person experiences a host of debilitating symptoms, including hallucinations, delusions, and paranoia. These symptoms can be disabling, and can exclude schizophrenic people from engaging with society. Only 10% of schizophrenic people are in employment, and it reduces life expectancy by 20 years.
The cause of schizophrenia is still a mystery. It’s difficult to treat an illness that you don’t understand, and many patients find the available medications give severe side effects, or that they barely help at all. To create better treatments, we need to understand how schizophrenia works.
To help us, there is one lead we can follow. It has been observed time and again that schizophrenia runs in families. This suggests that there is a genetic component to the illness, since we share genes with our family, and pass them to our children. Genes are codes in your DNA, which create all biological machinery within our body. Every gene has multiple variants – the same gene, but with slightly different coding. For example, different variants of the same gene code for either blue, or brown eyes. Some variants can be ‘defective’, and can contribute to a person developing certain diseases. Since schizophrenia runs in families, it is possible that they are all inheriting the same defective gene.
Recently in the United States, large numbers of these families were found. They live in Pennsylvania, and are part of the Amish people.
The Amish are a traditionalist sect of Christianity, with communities living in isolation. In closed societies, couples are more likely to have shared ancestry, since few outside people ever enter these circles. If there is a defective gene in the family, over time the number of offspring with this variant can increase. In the case of the Amish, this created large families with schizophrenia running down the generations.
Studying these Amish schizophrenics, researchers found defective variants of a particular gene in many of these people – zfhx3.
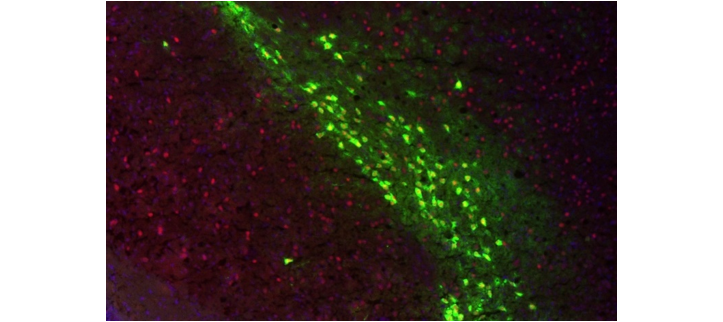
Microscopy image showing ZF activity (red) in dopamine neurons (green) in a mouse brain
This gene has recently risen to prominence, and is being increasingly implicated in a range of disorders. It is part of a family of genes whose only function is controlling other genes. Like a puppet master, it pulls strings across your DNA, controlling the activity of a wide range of other genes.
My group has been researching this gene for years – we call it ZF. We study the defective genes behind psychiatric diseases, using mice. This is because mice and humans are 99% genetically alike, making mice perfect subjects for studying genes behind human disease.
The news about defective ZF variants found in the Amish gave us an idea. Perhaps ZF is controlling genes that are active in the brain – and if we find which genes, maybe we can discover more about schizophrenia. This is where my project began, and my first task was to see exactly where in the brain ZF is working.
I looked at mouse brains under the microscope, using a marker that illuminates areas of ZF activity. I saw that ZF was active in specific areas of the brain that are rich in dopamine – a signalling molecule that has important roles in learning and pleasure. This was big news, as it is widely thought that dopamine malfunction plays a role in schizophrenia.
I had found a link: ZF could be controlling the genes in these dopamine-rich regions, and its dysfunction could lead to schizophrenia.
To test this, we created a colony of altered mice. Using cutting edge techniques, we deleted the ZF gene out of their DNA. More precisely, we deleted the gene only in these dopamine-rich regions of the brain. This means we can look at the raw influence of ZF in these regions, and figure out exactly why it’s needed there. Without ZF working in these dopamine-rich regions, would we make schizophrenic mice?
Truthfully, a mouse cannot have schizophrenia. Their brains are smaller and simpler, and mice do not have the cognitive power to experience the symptoms we see in humans. However, we can break down psychiatric illnesses into simpler behaviours, and test the mice on these. I do this by introducing them to environments that are designed to elicit a response, and measuring their reactions.
I have been studying these mice for 2 years, and their behaviour is truly puzzling. They are apathetic both to their own safety, and to new experiences. They sleep more often, but for less time. They over-react to sound, but under-react to light. They are highly aggressive with each other, with some mice having to live separately, however they are docile with humans.
I have also studied the brain structures of these mice, to try and gain insight into what could be causing these behaviours. Remember that ZF controls other genes – in these brains, I found significant defects in the signalling system of the dopamine-rich regions. This suggests that ZF controls the activity of the genes that compose this signalling system. With this system broken, brain cells are unable to communicate as they should, which may explain the erratic behaviour of these mice.
I am far from finished from studying ZF’s role in schizophrenia. I will be continuing to study the behaviour and brain structures of these mice, until we have a more complete picture of everything ZF does in these dopamine-rich brain regions.
This powerful gene has yet to give up its secrets, and schizophrenia may be one of them. If we conclude that that ZF dysfunction is causing schizophrenic symptoms, this could make ZF a target for future drug development – a precision drug, which could repair the damage caused by ZF dysfunction. While this won’t stop inheritance of schizophrenia, it could allow patients to be free of their symptoms, and hear no more phantoms knocking at the door.
Winner of the 2021 Max Perutz Science Writing Award
Max Perutz Science Writing Award 2021 shortlisted articles