Researchers at the MRC Harwell Institute have gained new insights into the function of the gene Katnal1. Katnal1 is one of a small family of genes that have been linked with intellectual disability, autism and schizophrenia in humans.
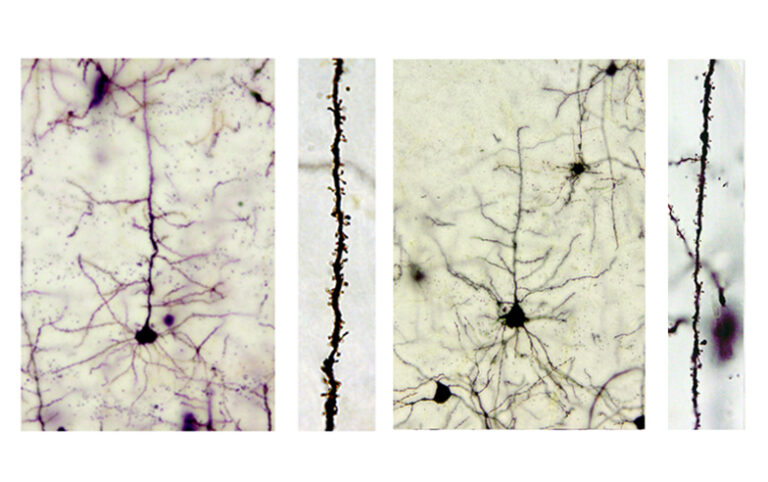
In mice, loss of function of the gene leads to poor learning and memory while the growth, migration and shape of neurons in the brain are all disturbed. This research highlights Katnal1 as a prime candidate for further study of the mechanisms underlying diseases of cognitive dysfunction. We have approximately one billion nerve cells in our brain. These neurons form a complex architecture of networks, which communicate with each other and with other areas of the body through chemical signals. Very early in development, neurons migrate from their birthplace to their final destination in the brain. During this period they develop and form numerous elaborate branches enabling crucial connections to be made with many other neurons. Defects in these processes have been associated with many cognitive disorders. The image shows neurons in normal mice (left) and mutant mice (right). In the mutant mice it can be seen that the neuron branches are shorter and thinner. The gene Katnal1 codes for a protein which determines the shape of microtubule structures within cells. In neurons, microtubules are important for directing neuronal migration and branching. Katnal1 and its family of genes enable the reshaping of microtubule structures at the appropriate time in developing neurons and the termination of branch growth so new ones can be formed. This gene previously has not been well characterised, although in a small patient study, loss of the gene was related to intellectual disability while one rare gene alteration has been linked to schizophrenia. In this study, mice with a coding sequence error in Katnal1 were identified as part of a large scale genetic study. The error, or mutation, resulted in a non-functional gene – it was essentially ‘switched off’. When the behaviour of the (mutant) mice was compared to normal mice (with the correct gene) a range of behavioural abnormalities were seen, including poor learning and memory. Changes in the brain, detectable only at a microscopic level, seemed to underlie these behavioural disturbances. Analysis of different brain sections showed that the patterns of neurons in the hippocampus (a region of the brain associated with memory) and cortex (the outermost layer of the brain) were different in mutants. The cortex has well defined cell layers so anomalies are easy to spot. More neurons were seen in the outer layers of the cortex in mutants, suggesting that the neurons may have migrated too far. Furthermore the neurons from mutants had a different shape and fewer synaptic spines – these are the structures on neurons that enable communication with other neurons. Defects were also seen in the cilia of the mutant mice. Cilia are hair-like protrusions that stick out from all cells and are vital in early development. In the brain, cilia are thought to maintain the circulation of chemicals and nutrients in the cerebrospinal fluid – a colourless fluid which maintains a healthy environment for the brain and its neurons. Defective cilia have been linked to many brain disorders including intellectual disability. Dr Pat Nolan, one of the authors on the paper, commented: “Our findings highlight the importance of this small group of genes in establishing the neuronal connections that are critical for precise brain functions” Further study of this gene and its role in neuron growth and development may provide insight into the cognitive dysfunction underlying intellectual disability and conditions such as autism. This will increase the likelihood of being able to identify therapeutic targets and potential treatments in the future. To read the research in Molecular Psychiatry click here