The Mary Lyon Centre (MLC) has pioneered the development and use of home cage monitoring (HCM) systems for the phenotyping of mice, to improve both the sensitivity and accuracy of observations and to improve the welfare of the animals by reducing handling and operator-induced stress.
Over the last few years, our team led by Sonia Bains has developed and tested the system and has obtained significant success in identifying a number of subtle and early onset phenotypes in wild-type strains and mutants alike. Some examples are the analysis of individual mouse activity in group-housed animals, the assessment of mouse behaviour throughout the dark/light cycle, and the use of vocalisation recordings in the HCM system.
The initial project, funded by a CRACK-IT challenge grant from the National Centre for the Replacement Reduction and Refinement of Animal Research (NC3Rs), led to a citizen science project called Rodent Little Brother in which thousands of members of the public contributed to the observation and analysis of the videos generated by the system, resulting in faster and non-biased data resolution.
The work carried out on the HCM systems at the MLC is at the heart of a COST Action that was funded by the European Commission in 2021. The Teatime COST Action aims to extend the use of HCM systems by sharing information and data with an expanding network of scientists from 27 countries.
Over the last few years, Sonia and her collaborators have been working on developing an algorithm to automatically annotate behaviours using the Home Cage Analyser by Actual Analytics. The ultimate goal is to extract vast amounts of data from a single experiment by harnessing the power of automation, and to describe phenotypes in both wild-type and mutant mouse strains. Sonia’s work showcasing the automated annotation of climbing behaviour was recently published in Frontiers in Behavioural Neuroscience. The paper describes how the use of the HCA system to assess climbing behaviour has led to the identification of a subtle but clear sexual dimorphism and a decline in activity with age in wild-type mice. When the method was applied to a mutant mouse line, it led to the early detection of motor impairment in the N171-82Q mouse model of Huntington’s disease.
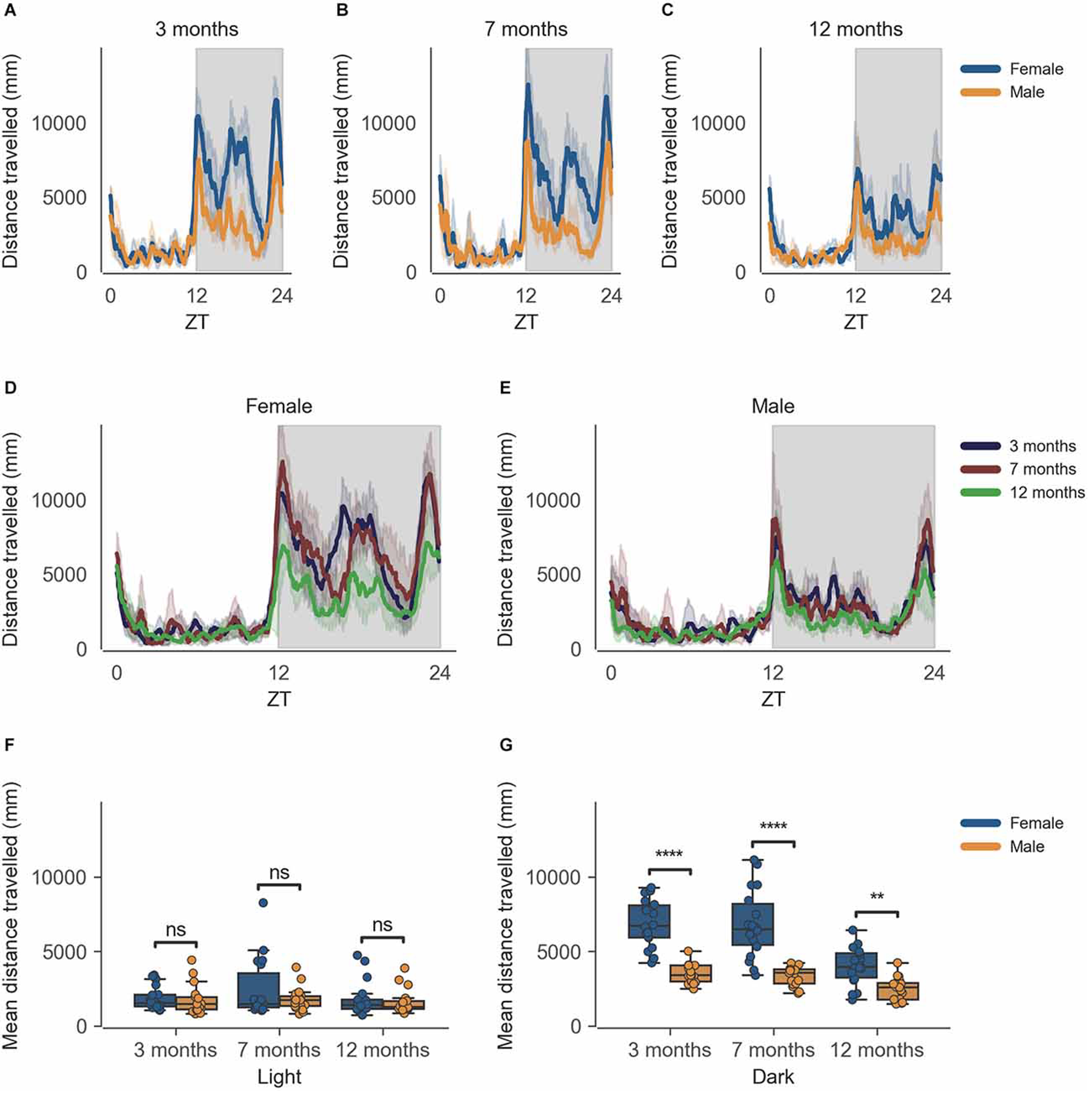
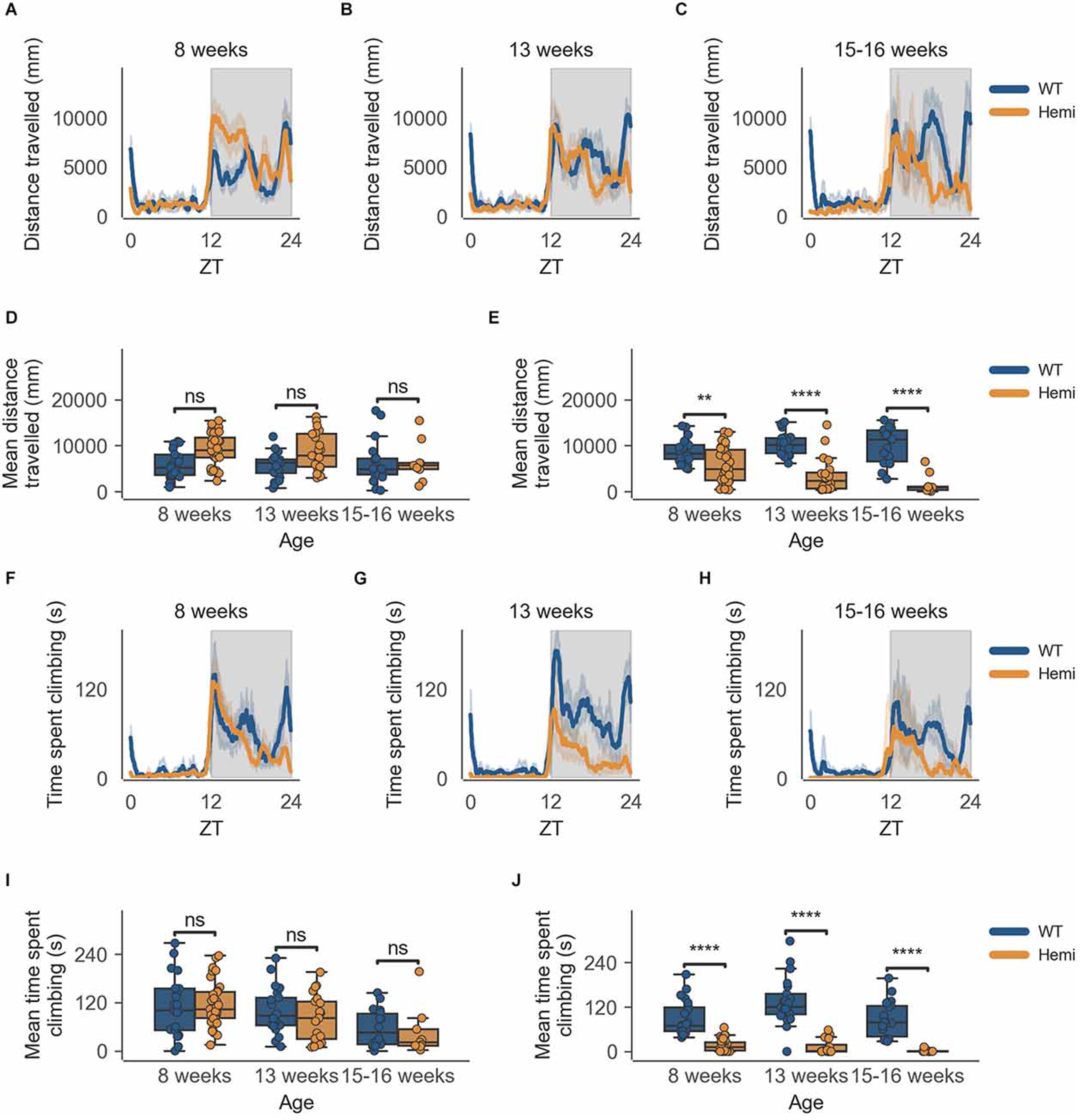
Aside from the interesting and important scientific advances provided by this project in assessing neurological decline through motor activity without operator intervention and observation bias, this paper provides further evidence that HCM systems are a positive development both for science and for animal welfare, and that the two go hand in hand.